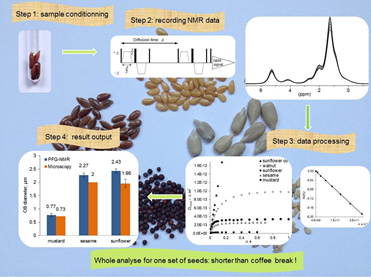
In oleaginous seeds, the energy reserves are mainly triacylglycerols that are liquid lipids contained inside micrometric vesicles, called oil bodies (OB). Seeds are of first importance for human and animal feeds, for biofuels sources and their economic weight is far from being negligible. Therefore a rapid and reliable method to measure the size of oil bodies is of first importance not only for biological aspects but for industrial reasons too. As a matter of fact the energy required to extract the lipids depends strongly on the size of the oil bodies. Up to now such measurements have been obtained by optical and electronic microscopy techniques, which are undoubtedly efficient but known as time consuming approaches (sample fixation, sectioning, staining and meaningful data collecting). LRM and PCI laboratories have been involved for a long time in methodological development and applications of Pulsed Field Gradients in Nuclear Magnetic Resonance (PFGNMR). PFGNMR has been demonstrated to be an appropriate and powerful tool to measure self-diffusion coefficients of molecules. As a matter of fact these methods have been successfully applied to study transport properties in solutions, complex fluids (cosmetic products), polymer membranes and biological systems. If molecular motions are restricted, which means that molecules face physical barriers during the time of the experiment, the analysis of NMR data leads to measurement of the size of the volume in which molecules are confined.
In this work we demonstrate for the first time that a standard liquid-phase NMR spectrometer equipped with a weak-intensity gradient coil can be used to determine the mean size of seed OB. The average diameters of OB, measured for several seeds by PFGNMR, are fully consistent with the previously published values obtained by microscopy techniques. The main advantage of the NMR method is that it does not require any sample preparation, experiments are performed with seeds directly introduced in standard NMR tubes and seeds are not altered by the experiment. The NMR approach also gives at the same time the total amount of TAG in seeds. A breakthrough of this
in vivo approach has been to evidence a TAG transfer through the network of interconnected OBs, which is dependent of the ability of adjacent membranes to open diffusion routes between OBs.